 
Consequently, our understanding of organic chemistry. Its atomic number (Z) is 6, therefore its nucleus is composed of. To get those two additional electrons, it can accept two electrons from somewhere else, or it can share two of its electrons with some other atom or atoms. Four elements, hydrogen, carbon, oxygen and nitrogen, are the major components of most organic compounds. Carbon belongs to period 2 and group 14 in the periodic table. An oxygen atom has six valence electrons it needs two more to achieve a stable electron configuration. Carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. 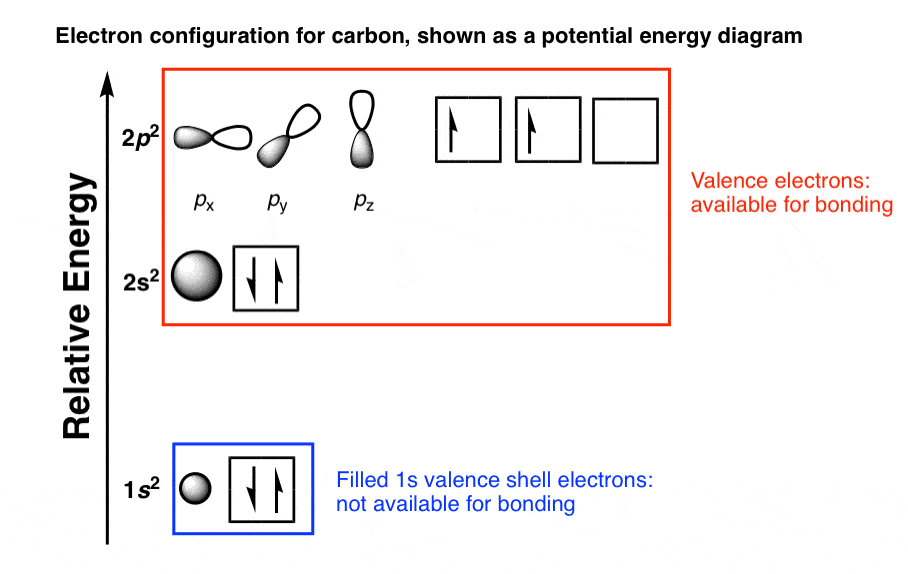
What happens when an oxygen atom only has other oxygen atoms to interact with? If a carbon atom is not going to lose four electrons to form a positively charged ion with a stable electron configuration, then an oxygen atom is certainly not going to lose six electrons. This is why covalent bonds are so strong.Īn oxygen atom will easily form an O 2- ion, but it needs some other atom or atoms to supply it with two electrons. The ground state electron configuration of ground state gaseous neutral carbon is He.2s2.2p2. The region between two positively charged atomic nuclei is incredibly stable for a negatively charged electron. Carbon atoms have 6 electrons and the shell structure is 2.4. A distinctive feature of the next atom is. The carbon atom has a total of 4 valence electrons which can be covalently bonded to similar atoms or other atoms. This will determine the ability of the carbon atoms to form long C chains. 
Chemical bonds formed between atoms by the sharing of electrons are called covalent bonds (“co-” means shared and “-valent” refers to valence electrons… shared valence electrons). Period 1: filling K shell (1st energy level), Z1. Atomic number, 6, Relative atomic mass, 12.011 State at 20C, Solid, Key isotopes C, 13C, 14C Electron configuration, He 2s22p CAS number, . For its covalent bonds, the carbon atom has a unique characteristic with atomic number 6 electron configuration. If the carbon atom shares its remaining three valence electrons with three other hydrogen atoms, all five atoms can achieve stable electron configurations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
